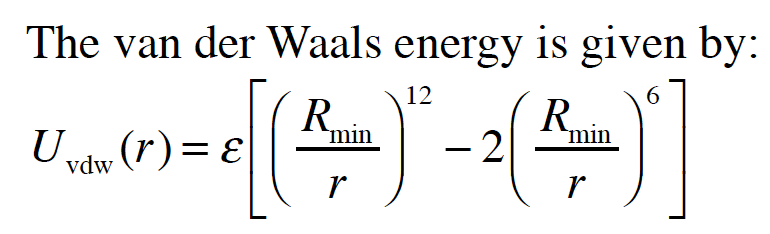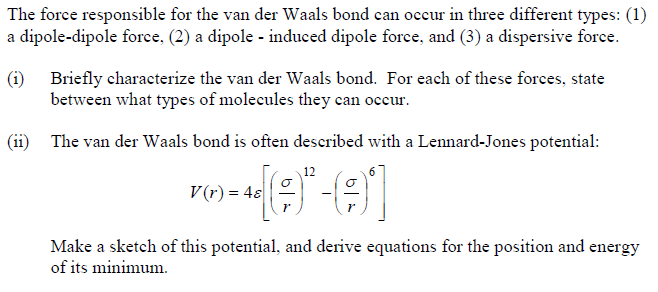Why is 'You cannot apply van der Waals equation for ideal gases' true? My teacher says it is true, but I think you actually can. - Quora

MathType - Van der Waals forces include attractions and repulsions between atoms, molecules, and surfaces, as well as other intermolecular forces. It is actually a weak force that results from a transient

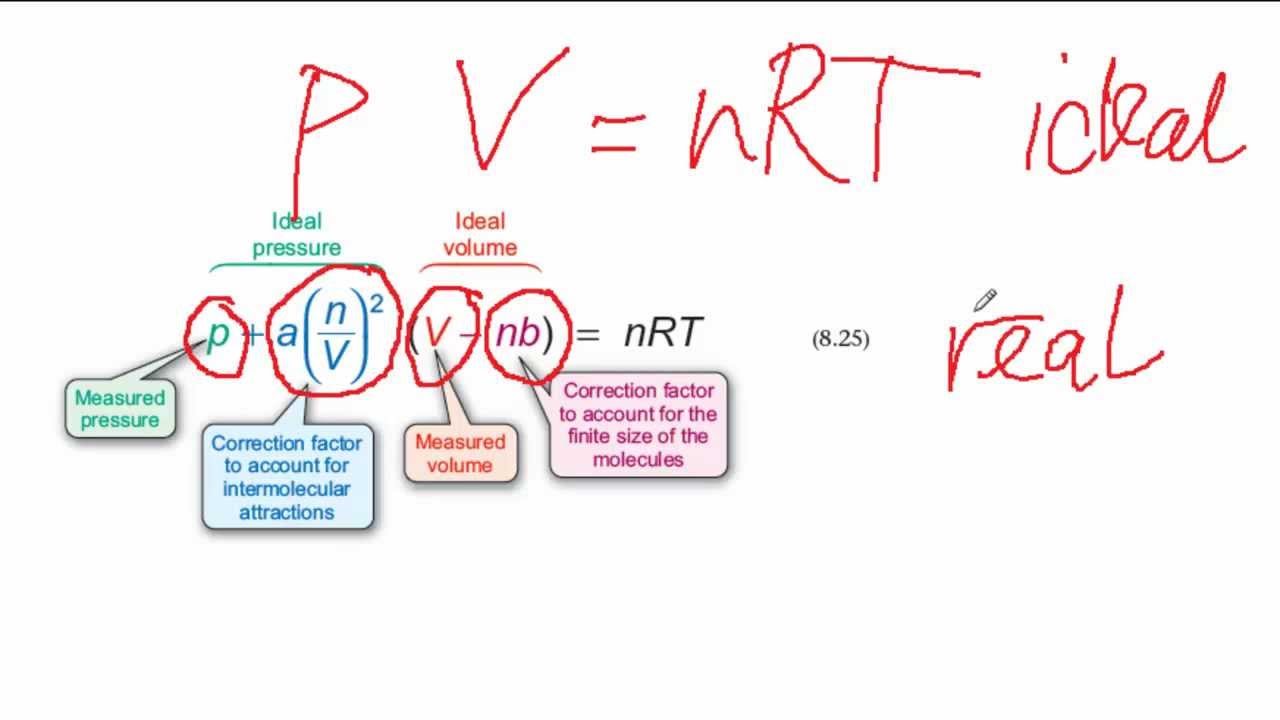
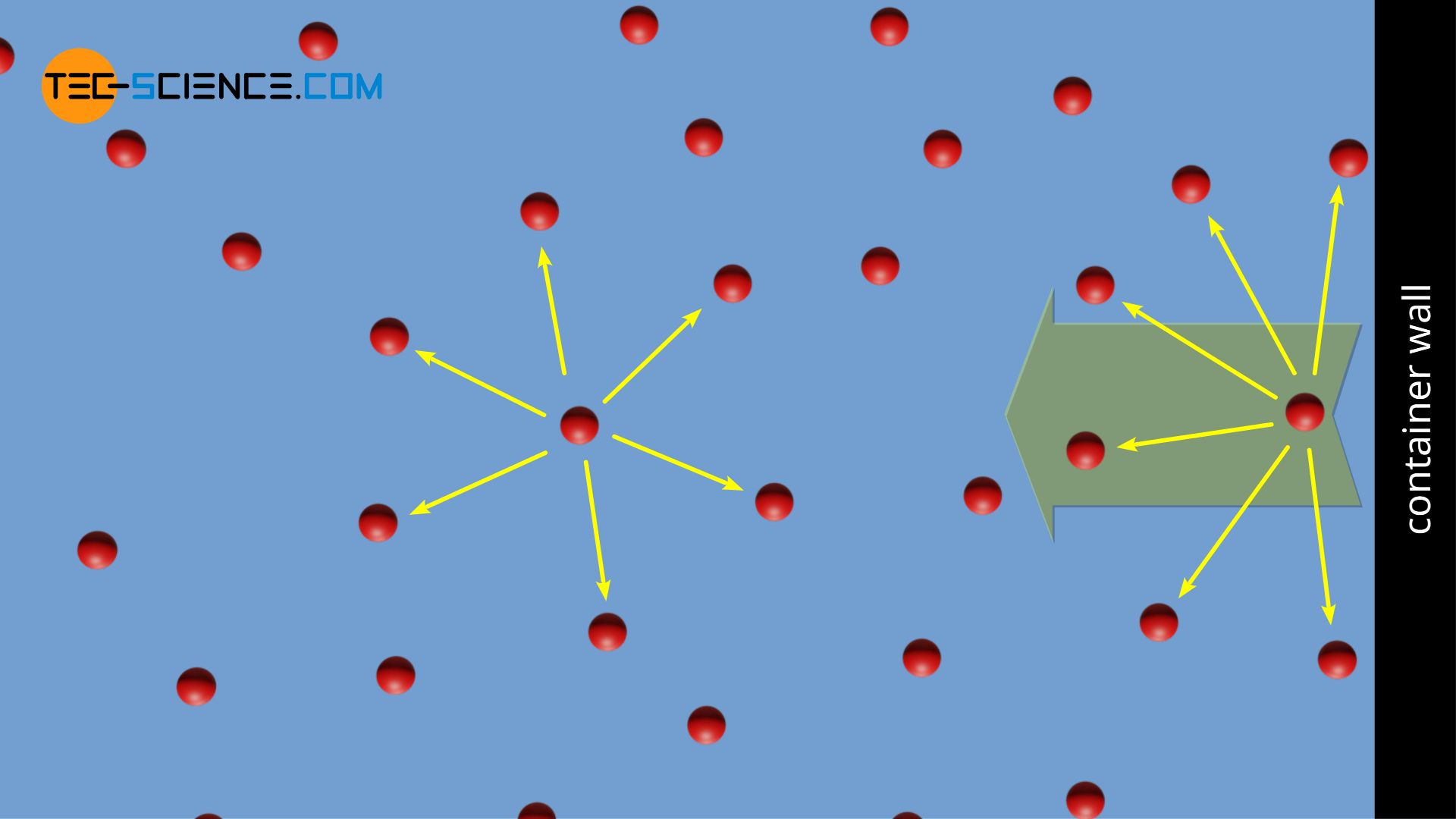
ReasonThe kinetic theory postulates of negligible volume of gaseous molecules and intermolecular forces of attraction do not stand correct high pressure and low temperature.AssertionVan der Waals equation describes the behaviour of real