Osmosis, osmotic pressure, laws of osmotic pressure and theories of osmosis presentation lecture slides for chemistry and pharmacy students | PPT

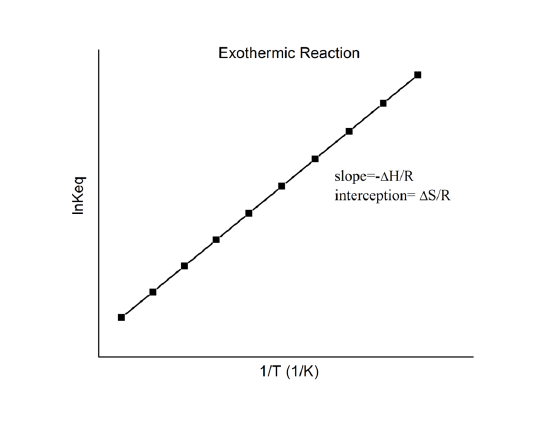
p 2.104 Van't Hoff - Boyle's law Il be of dissolved molecules is considered to be analogous to pas molecule could be considered to be due to be mandment of solute particles
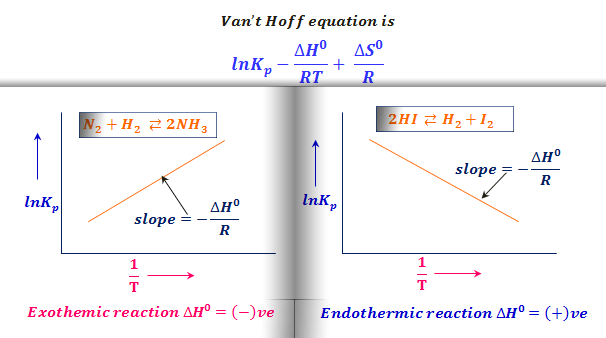
Van't Hoff equation-equilibrium. How much Van't Hoff equation — effect… | by Chemistry Topics | Medium

SOLVED: TC = g C o RT van't Hoff equation Osmotic pressure (atm) of particles per mole solution (Osm mol) Concentration (mmol) Reflection coefficient (varies) from Gas constant 0.082 atm mol Absolute
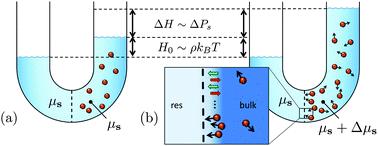
Van't Hoff's law for active suspensions: the role of the solvent chemical potential - Soft Matter (RSC Publishing)




![D] Laws of Osmotic Pressure and van't Hoff's' Equation van't Hoff (1877).. D] Laws of Osmotic Pressure and van't Hoff's' Equation van't Hoff (1877)..](https://classroom-images.cdn.askfilo.com/classroom/1668316067828_fpynsrpl_3026990.jpg)